Battling bacteria with viruses
New agents to fight multidrug-resistant germs
Advertisement
Resistance to antibiotics is on the rise worldwide. Fraunhofer scientists have joined forces with partners in the Phage4Cure project to explore alternatives to antibiotics. One objective is to vanquish multidrug-resistant pathogens with viruses called bacteriophages. Another is to see these phages approved to treat the dreaded hospital germ Pseudomonas aeruginosa, the most frequent bacterial cause of pneumonia.
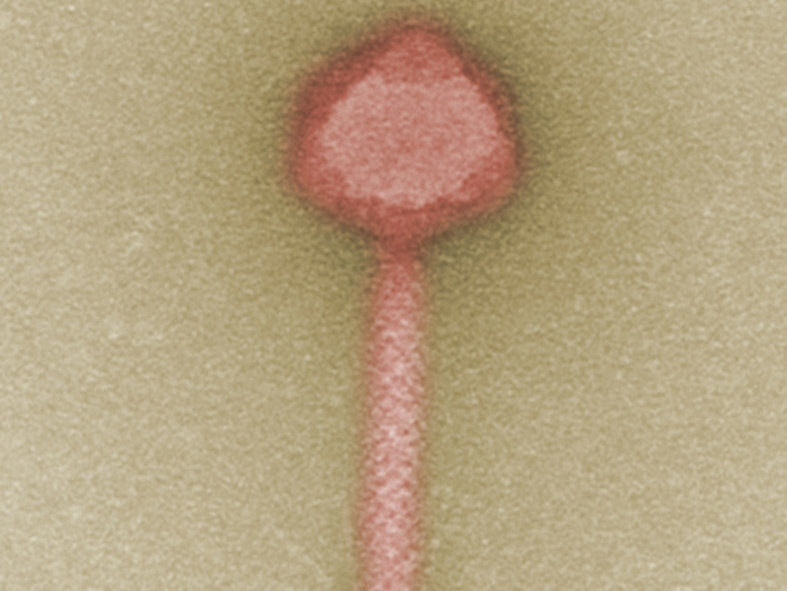
A candidate for future phage therapy - bacteriophage against multidrug-resistant clinical strains of Pseudomonas bacteria.
© M. Rohde/HZI Braunschweig
According to the World Health Organization (WHO), antibiotic resistance has reached alarming levels worldwide. WHO Secretary General Tedros Adhanom Ghebreyesus says it is jeopardizing a hundred years of medical progress. Finding a solution to this problem is one of the most pressing challenges in healthcare. This is where the Phage4Cure project enters the picture. Partners in this project, including the Fraunhofer Institute for Toxicology and Experimental Medicine ITEM at Braunschweig, aim to enlist bacterial viruses, also known as bacteriophages or simply phages, to wage war on bacterial infections. Phages are viruses that invade and multiply in bacteria, finally causing them to lyse, or burst. The great advantage of phages is that they attack only a specific host bacterium, ignoring bodily cells and other bacteria. In Germany, phage preparations have not yet received regulatory approval, which is why the Phage4Cure partners are seeking to establish phages as a new drug. “Phage therapy is nothing new; countries of the former Soviet Union have used it successfully for decades. However, this kind of personalized treatment has not yet been approved in Germany, a major reason being the lack of clinical trials. But now, in light of antibiotic resistance, more and more research is focusing on phages – especially since no new antibiotics are being developed by the pharmaceutical industry,” says Prof. Holger Ziehr, project manager at Fraunhofer ITEM.
Phages as an additional therapeutic approach
The partners’ efforts towards a new phage therapy address every step of the development chain: the selection of promising phages, development of the manufacturing process for the phage as a pharmaceutically active agent, manufacturing of the investigational medicinal product, preclinical research and clinical trials. The first item on their agenda is to develop an inhalable drug cocktail comprised of three bacteriophages to treat for the multidrug-resistant bacterium Pseudomonas aeruginosa, the most common bacterial cause of pneumonia in cystic fibrosis patients and urinary tract infections. It can even lead to septicemia, or blood poisoning. The new phage cocktail will have to comply with European pharmaceutical requirements. Cystic fibrosis patients are intended to be the first to benefit. Many more could follow. “Our goal is to develop phages as an additional therapeutic approach for a variety of infectious diseases, in particular those where antibiotics are no longer effective,” says Ziehr.
Fraunhofer ITEM in Braunschweig is responsible for the manufacturing process, which involves the propagation of Pseudomonas aeruginosa cells in bioreactors. “Once a certain cell density has been attained, we add phages that invade the cells and continue to propagate there until all bacteria are destroyed. This leaves a clear culture broth containing phages, which we purify pharmaceutically in the next step,” says Ziehr, a biochemist. The manufacturing process is platform-like by design. With a few minor adjustments, it can be adapted to other phage manufacturing processes. Fraunhofer ITEM sources phages from the DSMZ. (See the facts-and-figures box for details.)
Mixing a potent phage cocktail
The idea here is to combine three different phages in a cocktail for the patient. Collecting clinical bacterial isolates from cystic fibrosis patients was an early priority on the project’s to-do list. The DSMZ then identified phages that are able to dissolve the isolates. “This screening pinpointed three phages with the broadest possible host spectrum. Collectively, they can lyse 70 percent of the 150 isolates. We would be able to heal around 70 of 100 patients with our phage cocktail,” says Ziehr.
Fraunhofer ITEM has been GMP-licensed to produce biologics such as proteins for more than 20 years. It is now going to apply for an extension to the manufacturing license to include phages as well. Once this extension will have been granted, the institute will be in a position to produce the three phages and mix them to a cocktail that will serve as investigational medicinal product for clinical trials. Preclinical research is scheduled to start in spring at the Fraunhofer ITEM’s Hannover site. “Two more phage projects have been added since Phage4Cure. In the PhagoFlow project, we are developing bacterio-phages to rapidly treat sepsis in wounds. In the Phage2Go project, we are developing phages for inhaled MRSA therapy,” says Ziehr, shedding light on further projects to research treatments for bacterial infections.



















































