New insights into regulation of food intake in mammals
New therapeutic approaches for obesity and diabetes
Advertisement
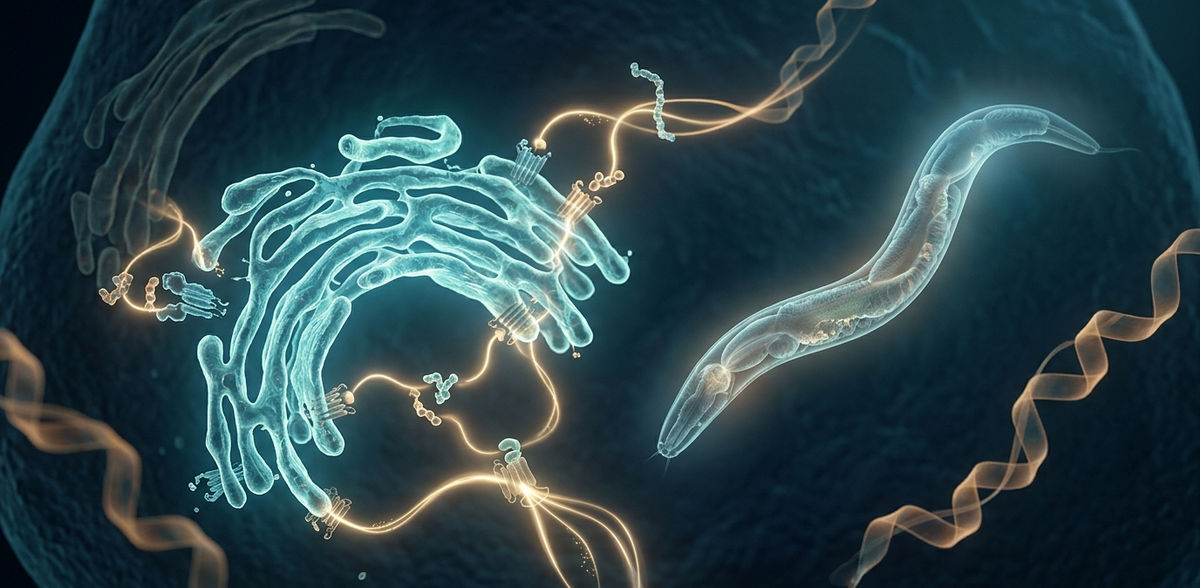
An international team, including scientists from Leipzig University, has gained important new insights into the regulation of food intake in mammals. The study, recently published in the renowned scientific journal PNAS, shows that the relative availability of saturated and monounsaturated fatty acids within the cell’s branched membrane system – the endoplasmic reticulum (ER) – plays a central role in regulating food intake. The researchers also identified a potential genetic precursor of the GLP-1R/GIPR receptor group. This could open up new avenues for the development of therapies for obesity and metabolic disorders.
The study investigated food intake in the nematode C. elegans, providing new evolutionary insights into the regulatory process. This organism is widely used in research to study development and genetics. Notably, unlike mammals, it lacks both the hormone leptin and leptin receptors involved in the regulation of food intake. Food intake is regulated via the endoplasmic reticulum (ER) – through a balance between saturated and monounsaturated fatty acids. This process is activated by the stress sensor IRE-1 (inositol-requiring enzyme 1), located in the ER membrane, which influences behaviour via neuronal serotonin and the G protein-coupled ligand–receptor pair PDF-1/PDFR-1 – a signalling pair involved in cellular communication and the regulation of specific physiological processes. This mechanism triggers both hedonic signals – associated with the pleasurable aspects of eating – and homeostatic signals, which reflect the body’s physiological needs.
These two types of signalling interact to regulate food intake. The study shows that this system is homologous to GLP-1/GIP-related systems in mammals, which play an important role in regulating blood glucose levels and food intake. While the PDF-1/PDFR-1 signalling pair has only a modest effect, it helps, in mice for example, reduce body weight and improve blood glucose control. This may prove promising for future treatments of obesity and diabetes.
Collaboration between the researchers in this field was initiated in 2024, when Professor Ronald Kahn of Harvard Medical School and Professor Annette Beck-Sickinger of Leipzig University jointly took part in the international symposium “Obesity Mechanism”, organised by Collaborative Research Centre 1052 at Leipzig University’s Faculty of Medicine. The project was led by researchers at the Joslin Diabetes Center in Boston and Harvard University in Cambridge, Massachusetts, who were responsible for the overall study design and carried out the key in vivo experiments in nematodes and mice. They were supported by several renowned universities and research institutes in the US, China and Japan. In Leipzig, the early career researcher Hannah Lentschat, working in Professor Annette Beck-Sickinger’s group, developed, synthesised and tested the peptides investigated in the study – small protein molecules that play a central role in metabolic regulation. The group also carried out key in vitro experiments, which provided valuable insights into the mode of action of these substances.
The findings of the study will now be further pursued within the Leipzig Center of Metabolism (LeiCeM) Cluster of Excellence – a central research hub for metabolic diseases at Leipzig University. Collaborative Research Centre 1423, Structural Dynamics of GPCR Activation and Signaling, not only supported the collaboration in terms of expertise and infrastructure, but also provided essential resources, particularly for the complex peptide syntheses. “This study presents a new paradigm for understanding appetite regulation in response to metabolic signals. Our findings suggest that food intake is governed by a complex interplay of hedonic and physiological signals,” says Professor Annette Beck-Sickinger, spokesperson of CRC 1423 and one of the study’s lead authors.
The researchers plan to build on these findings in further studies to elucidate the precise molecular mechanisms and evolutionary parallels between nematodes and mammals. This could lead to new therapies for obesity and metabolic disorders based on the regulation of food intake through metabolic and neuronal signals.