Researchers engineer hardier microbes to improve bioproduction of fuels, chemicals
Advertisement
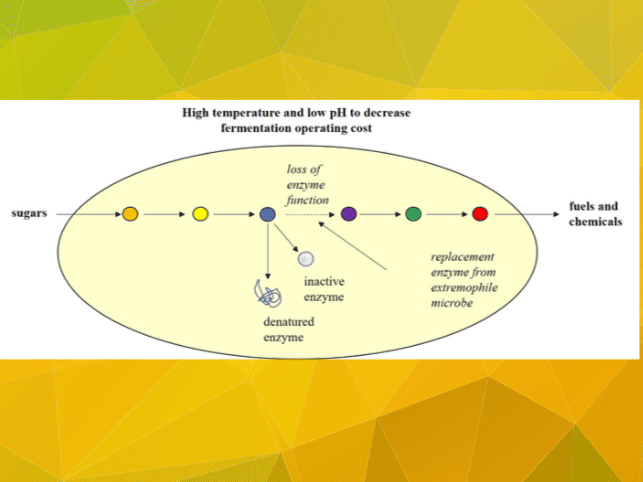
Busy, productive microbes use enzymes to break down leaves, stalks and other biomass and then convert that material into renewable fuels and chemicals. But some of those enzymes can’t function at the high temperatures or acidity that maintain low operating costs for fermentation processes.
Researchers are working to identify and produce robust enzymes that can replace other enzymes that break down in the fermentation of bio-based fuels and chemicals.
Laura Jarboe
A research team led by Iowa State University’s Laura Jarboe will try to fix that problem by identifying rougher, tougher enzymes and engineering microbes to use them in industrial fermentation. Their work is supported by a three-year, $969,000 grant from the U.S. Department of Energy.
“We want to make these microbes more robust,” said Jarboe, Iowa State’s Cargill Professor of Chemical Engineering. “To do that, we need to think about the problem for the microbes. We can’t just say, ‘Do better.’”
In this case, enzymes that require cooling or pH (acidity/alkalinity) adjustments to remain active are costly problems. Some of these enzymes are vital to the microbes’ ability to convert bio-based substrates into products, and some are vital for organism survival.
Are there hardier, tougher, more robust enzymes out there? Could industrial microbes be engineered to be more like “extremophiles” and remain active under the harsh conditions in the fermentors?
Teaming up to find answers are Jarboe; Robert Jernigan, an Iowa State Charles F. Curtiss Distinguished Professor in Agriculture and Life Sciences in the Roy J. Carver Department of Biochemistry, Biophysics and Molecular Biology; and Peter St. John, a senior scientist in the Biosciences Center at the U.S. Department of Energy’s National Renewable Energy Laboratory in Golden, Colorado.
Millions to study microbes
The energy department’s Office of Biological and Environmental Research awarded 34 biotechnology grants totaling $45.5 million this summer, including this grant supporting the work of Jarboe, Jernigan and St. John. Winners were selected after a competitive, peer-review process.
“Biofuels that can power planes and ships, and bioproducts made from renewable resources will play a critical role in decarbonizing our economy,” said Secretary of Energy Jennifer M. Granholm, in a statement announcing the grants.
The energy department’s research program is targeting two areas of study: One, re-engineering microbes that help convert biomass and synthetic polymers into fuels and products. And two, developing imaging technologies to better study the plants and microbes used to produce bioproducts.
The various projects “will help us understand, predict, and even design (biofuels and bioproducts) at the cellular level, so that we can unlock their full potential,” Granholm said.
Labs to factories
Jarboe, Jernigan and St. John have developed a three-part plan to reengineer microbes so they produce heat- and acid-resistant enzymes:
- St. John will lead efforts to build computational, genome-scale models of microbial metabolism, including the chemical reactions that convert sugars into energy. The models will help the researchers predict how higher temperatures or changes in acidity will change the reactions. “Our work will try to identify which enzymes react to these stresses and adversely affect metabolism, which can then be targeted for replacement,” he said.
- Jernigan will lead searches of enzyme data to identify hardy replacements for the enzymes that fail at high temperature or acidity. “I’ve studied proteins all of my life and we have incredibly rich data – several hundred million protein sequences – and this is an opportunity to make practical applications of this information,” he said.
- Jarboe will lead efforts to engineer microbes that can produce the robust replacement enzymes. There will also be experiments to evaluate and characterize the microbes, enzymes and resulting chemical reactions. “Maybe it doesn’t matter that some of the processes rely on highly sensitive enzymes,” she said. “Maybe there are other enzymes that can do the exact same job.”
If that’s the case, those replacement enzymes could one day make a difference in how biorenewable fuels, chemicals and other products are manufactured.
“Finding a systematic approach to engineering thermotolerance and pH-tolerance in microbes,” St. John said, “will hopefully allow newly developed strains to move from the lab to industry with greater speed and lower cost.”






















































