Sleeping Beauty enables efficient gene transfer in haematopoietic stem cells of humans
Advertisement
gene therapy of haematopoietic stem cells aims at a life-long therapeutically effective correction of these cells in humans with particular genetic disorders. This therapeutic approach has proved to be effective in clinical trials while the risk of leukaemia development as a side-effect of the therapy must be kept as low as possible. In an international research network, researchers of the Paul-Ehrlich-Institut have developed a method using "jumping genes" (transposons) to transfer genes efficiently and to anchor them in the genome of the modified cells.
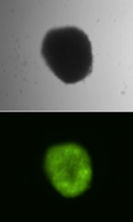
Stable gene transfer and expression of a Venus fluorescence marker gene mediated by Sleeping Beauty transposons in hematopoietic progenitor cells.
PEI
In gene therapy of certain congenital disorders, the therapeutic gene is transferred into the target cells using particular transport vehicles, also known as vectors. Gene transfer into haematopoietic (blood forming) stem cells has been successfully tested in clinical trials for the treatment of various genetic disorders caused by a defect in one particular gene (monogenetic). Examples include certain immune defects such as the group of Severe Combined Immune Deficiency disorders (SCID). In some studies, however, isolated cases of oncogenic mutations triggered by gene vector integration led to cancer (leukaemia) as an undesired side-effect of the therapy. Major efforts have ever since been undertaken to develop safer vector systems.
The research team of Dr Zoltán Ivics, head of Division Medical Biotechnology of the Paul-Ehrlich-Institut uses Sleeping Beauty for therapeutic gene transfer into human cells. The Sleeping Beauty (SB) transposon system is a non-viral vector, which is low-cost to manufacture, relatively simple to use. Up to now, its clinical use for hematopoietic stem cell therapy has been hampered by technical challenges.
In an international collaboration with other researchers from Germany, Spain and France, Ivics and his team have shown that gene constructs can be incorporated 20-times more efficiently into the genomes of hematopoietic stem cells if minicircle DNA is used to carry the SB transposon. Minicircle DNA is very small because sequences used for the manufacturing process of plasmids have been removed.
In addition, the efficacy and safety of the stable gene transfer ex vivo (outside the body) into the haematopoietic stem cells could be significantly increased by providing the transposase enzyme in the form of a synthetic messenger DNA (mRNA). This enzyme is required for the gene transfer.
"We have methodologically further developed the Sleeping Beauty transposon system in such a way that clinical use of this method is now possible," said Dr Ivics, as he described the potential use of these research results.
Original publication
Holstein M, Mesa-Nuñez C, Miskey C, Almarza E, Poletti V, Schmeer M, Grueso E, Ordóñez Flores JC, Kobelt D, Walther W, Aneja MK, Geiger J, Bonig HB, Izsvák Z, Schleef M, Rudolph C, Mavilio F, Bueren JA, Guenechea G and Ivics Z; "Efficient Non-Viral Gene Delivery into Human Hematopoietic Stem Cells by Minicircle Sleeping Beauty Transposon Vectors"; Mol Ther.; Published Online: January 17, 2018
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Gene therapy
Genetic diseases once considered untreatable are now at the center of innovative therapeutic approaches. Research and development of gene therapies in biotech and pharma aim to directly correct or replace defective or missing genes to combat disease at the molecular level. This revolutionary approach promises not only to treat symptoms, but to eliminate the cause of the disease itself.

Topic world Gene therapy
Genetic diseases once considered untreatable are now at the center of innovative therapeutic approaches. Research and development of gene therapies in biotech and pharma aim to directly correct or replace defective or missing genes to combat disease at the molecular level. This revolutionary approach promises not only to treat symptoms, but to eliminate the cause of the disease itself.




















































