Abnova and Clearbridge BioMedics Accelerate Circulating Tumor Cell System into the Research and In Vitro Diagnostic Markets
Advertisement
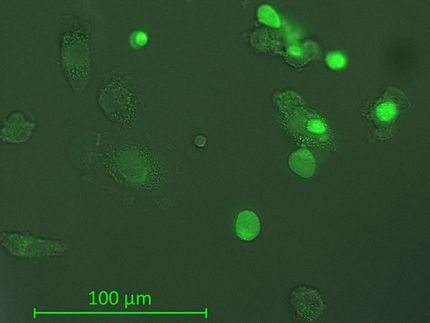
Abnova Corporation and Clearbridge BioMedics announced a collaboration to jointly develop a platform that provides a next generation, non-invasive “liquid biopsy” for cancer screening, staging, treatment, and monitoring. Through this collaboration, Abnova will leverage upon Clearbridge BioMedics’ core ClearCell™ CTChip®, which is an advanced microfiltration system for the isolation, enumeration, and retrieval of viable circulating tumor cells (CTCs) from blood. Abnova will then incorporate its proprietary antibody reagents (both research-grade antibody reagents and cGMP clinical-grade antibodies), to enable the accurate diagnosis of specific medical conditions or diseases.
“CTCs represent one of three important pillars of system integration for Abnova in the hierarchical organization and interrogation of tissue, cell, and protein,” said Dr. Wilber Huang, President and CEO of Abnova. “Clearbridge BioMedics has elegantly engineered a breakthrough CTC platform, which is unmatched by the competitors, specifically the ability to preserve cell viability after isolation. Up until now, the access to tissue samples is confined to the initial biopsy for diagnosis and staging workup followed by pathological confirmation of resected tissues. The opportunity for non-invasive “liquid biopsy” has tremendous potential in disease management, especially in molecular staging, personalized therapy and recurrence monitoring. Abnova has a plethora of research antibodies and FISH probes to catapult the research market, in addition to a full pipeline of novel IVD biomarkers under development, which are applicable to CTC platform including prostate, ovarian, and lung cancers."
Other news from the department research and development
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Antibodies
Antibodies are specialized molecules of our immune system that can specifically recognize and neutralize pathogens or foreign substances. Antibody research in biotech and pharma has recognized this natural defense potential and is working intensively to make it therapeutically useful. From monoclonal antibodies used against cancer or autoimmune diseases to antibody-drug conjugates that specifically transport drugs to disease cells - the possibilities are enormous

Topic world Antibodies
Antibodies are specialized molecules of our immune system that can specifically recognize and neutralize pathogens or foreign substances. Antibody research in biotech and pharma has recognized this natural defense potential and is working intensively to make it therapeutically useful. From monoclonal antibodies used against cancer or autoimmune diseases to antibody-drug conjugates that specifically transport drugs to disease cells - the possibilities are enormous