Decoding cellular zipcodes
Researchers suspect that neurodegenerative diseases occur when messenger RNA goes astray in the neuron
Advertisement
With a new method, Marina Chekulaeva is able to identify “zipcodes” that assign a destination to mRNAs. She has now presented her method in the journal Nature Neuroscience.
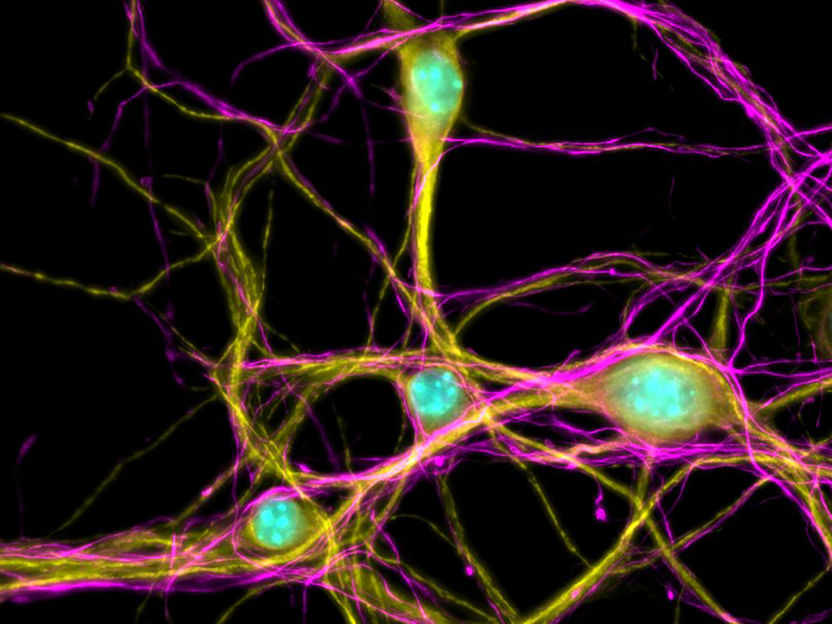
Primary cortical neuron
Samantha Mendonsa, MDC
In neurodegenerative diseases, the loss of neurons leads to a variety of malfunctions. In Alzheimer’s disease, for example, massive neuronal cell death in the brain causes the loss of cognitive abilities, while in amyotrophic lateral sclerosis (ALS) the death of motor neurons prevents commands from the brain reaching muscle cells in the body – resulting in progressive paralysis.
“Many of these diseases start with the loss of neuronal branches,” says Dr. Marina Chekulaeva, head of an independent research group at the Berlin Institute for Medical Systems Biology of the Max Delbrück Center (MDC-BIMSB). Chekulaeva believes that the mislocalization of messenger RNA molecules (mRNAs) is responsible for this loss. mRNAs deliver instructions for how to build important protein molecules – molecules that also play a key role in the interaction of neurons. So if mRNAs do not arrive at their correct location, the neurons lose a critical function: contact between neurons is lost, nerve tracts are interrupted, and the cells themselves die. A functioning mRNA “postal system” is therefore crucial for neurons to be able to do their important work.
“We know that cells contain hundreds to thousands of localized mRNA molecules,” says Chekulaeva. “So we can assume there are also mechanisms in place that deliver the mRNAs to their destination.” No one yet knows exactly how this cellular postal system works, but scientists have uncovered some pieces of the puzzle. In the 1990s, researchers in the US discovered a sequence at the tail of beta-actin mRNA molecule that functions like a zipcode. Transport molecules then act like the “postmen” of the cell, using these codes to assign a destination to the mRNAs. In the journal Nature Neuroscience, Chekulaeva’s team jointly with Ulitsky’s team at the Weizmann Institute of Science has now presented a method for identifying the zipcodes of additional mRNAs.
Mapping thousands of RNA sequences at once
Chekulaeva’s team determined the location of mRNAs in the cell and used a specially developed method to identify the sequences that play a role in their targeted delivery. “The special thing about our method is that it allows us to analyze thousands of RNA sequences simultaneously,” says Samantha Mendonsa, one of the first authors of the study. “This way, we can all at once identify multiple zipcodes that assign destinations to different localized mRNAs.”
Max Delbrück Center researchers have not only developed a method that allows them to identify a variety of previously unknown zip codes, they have also created a tool they can use to study the postal system of other polarized cell types in detail. “These zipcodes help us to decipher the entire postal system – with all the transport molecules and receivers that are required for localized mRNAs to reach their destination,” explains Chekulaeva.
This can also help to provide information about what goes wrong in the system of ALS patients, for example – i.e., why neuronal branches and eventually entire motor neurons are lost. Researchers hope this knowledge will ultimately contribute to a better understanding of this neurodegenerative disease, and even pave the way for new therapeutic approaches in the future.