Microscopy technique enables 3D super-resolution nanometre-scale imaging
Research team combine two techniques to achieve isotropic super-resolution imaging
Advertisement
Over the last two decades, microscopy has seen unprecedented advances in speed and resolution. However, cellular structures are essentially three-dimensional, and conventional super-resolution techniques often lack the necessary resolution in all three directions to capture details at a nanometer scale. A research team led by Göttingen University, including the University of Würzburg and the Center for Cancer Research in the US, investigated a super-resolution imaging technique that involves combining the advantages of two different methods to achieve the same resolution in all three dimensions; this is “isotropic” resolution. The results were published in Science Advances.
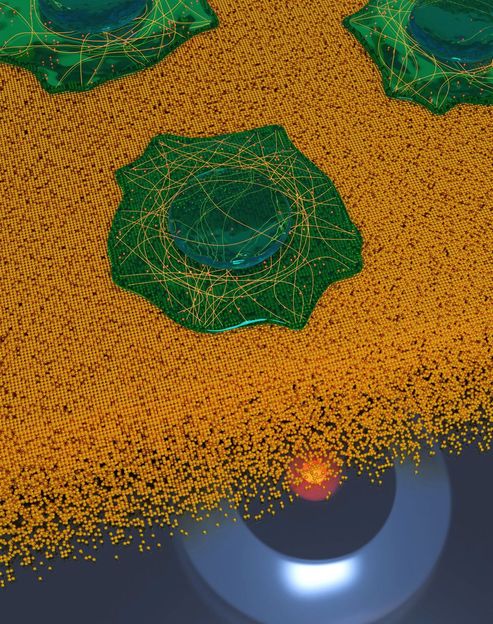
To show that 3D imaging with MIET-SMLM is compatible with biological samples, cells were seeded on a cover glass coated with 10 nm of gold and 5 nm of SiO2 using standard immunofluorescence sample prepa-ration procedure. The artistic rendering illustrates cells imaging on a gold surface resolving microtubules network and clathrin coated pits.
Alexey Chizhik
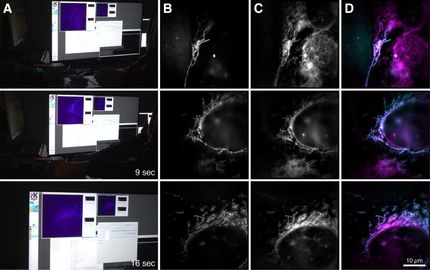
Despite tremendous improvements in microscopy, there still exists a remarkable gap between resolution in all three dimensions. One of the methods that can close this gap and achieves a resolution in the nanometer range is metal-induced energy transfer (MIET) imaging. The exceptional depth resolution of MIET imaging was combined with the extraordinary lateral resolution of single-molecule localization microscopy, in particular with a method called direct stochastic optical reconstruction microscopy (dSTORM). The novel technique based on this combination allows researchers to achieve isotropic three-dimensional super-resolution imaging of sub-cellular structures. In addition, the researchers implement dual-color MIET-dSTORM enabling them to image two different cellular structures in three dimensions, for example microtubules and clathrin coated pits – tiny structures within cells – that exist together in the same area.
“By combining the established concepts, we developed a new technique for super-resolution microscopy. Its main advantage is it enables extremely high resolution in three dimensions, despite using a relatively simple setup,” says Dr Jan Christoph Thiele, first author of the publication, Göttingen University. “This will be a powerful tool with numerous applications to resolve protein complexes and small organelles with sub-nanometer accuracy. Everyone who has access to confocal microscope technology with a fast laser scanner and fluorescence lifetime measurements capabilities should try this technique,” says Dr Oleksii Nevskyi, one of the corresponding authors.
“The beauty of the technique is its simplicity. This means that researchers around the world will be able to implement the technology into their microscopes quickly,” adds Professor Jörg Enderlein who led the research team at the Biophysics Institute, Göttingen University. This method shows promise to become a powerful tool for multiplexed 3D super-resolution microscopy with extraordinary high resolution and a variety of applications in structural biology.