New technique for in-cell distance determination
Advertisement
Researchers from the University of Konstanz, Bielefeld University and ETH Zurich demonstrate for the first time that the pulsed EPR technique RIDME (relaxation-induced dipolar modulation enhancement) can be used for in-cell distance determination in biomacromolecules. Applied within the cell, RIDME improves significantly on conventional double electron–electron resonance (DEER) measurements.
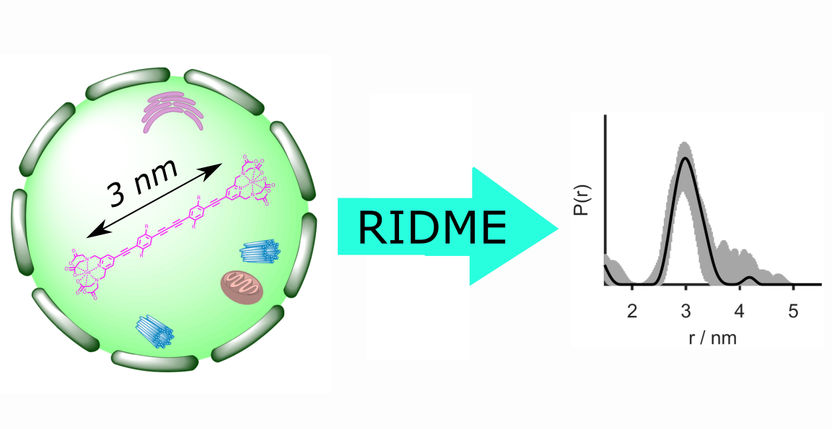
In-cell distance determination by EPR reveals essential structural information about biomacromolecules under native conditions. For the first time, the pulsed EPR technique RIDME (relaxation-induced dipolar modulation enhancement) was utilized for distance measurements inside cells. It provides a five-times improved sensitivity as compared to the previously used double electron-electron resonance approach.
Research group Professor Malte Drescher, University of Konstanz
In a joint paper which has just been published in The Journal of Physical Chemistry Letters, researchers from the University of Konstanz, Bielefeld University and ETH Zurich demonstrate for the first time that the electron paramagnetic resonance (EPR) technique RIDME (relaxation-induced dipolar modulation enhancement) can be applied to determine distances between gadolinium(III)-based spin labels in cells. In-cell distance determination by electron paramagnetic resonance (EPR) reveals essential structural information about biomacromolecules, including their conformation as well as folding and unfolding processes.
Conventional methods for in-cell determination of distances such as double electron–electron resonance (DEER or PELDOR) are principally much less sensitive than RIDME, provide up to five times smaller modulation depths, have certain limitations with regard to excitation bandwidth and are technically more demanding. As a single-frequency technique which makes use of relaxation-induced spin flips to determine the distance between two spin labels, i.e. between two unpaired electrons, RIDME overcomes all of these disadvantages.
What is special about this technique is that it allows the researchers to work with molecules under native conditions, as Professor Malte Drescher and lead author Dr Mykhailo Azarkh, both from the University of Konstanz, emphasize: “We started out by analysing the conformation of a protein inside the cell. With less sensitive techniques, we are forced to insert and tag a lot of protein to be able to observe it, which is not at all what happens in nature. Ideally, we want to be working with concentrations that are physiologically relevant. Since RIDME is much more sensitive than DEER, it allows us to do just that. We are now in a position to address issues that we would not otherwise be able to address”.
The performance of in-cell RIDME was assessed at Q-band using stiff molecular rulers labelled with Gd(III)-PyMTA and microjointed into Xenopus laevis (African clawed frog) oocytes. In other words, the researchers used a model system where the precise distance between the spin labels was already known, allowing them to verify the RIDME measurements.
In-cell RIDME distance determination was developed and tested as part of the on-going ERC-funded project “SPICE – Spectroscopy in cells”, for which Malte Drescher, Heisenberg Professor for Spectroscopy of Complex Systems at the University of Konstanz, and his research team were recognized with an ERC Consolidator Grant worth approximately two million euros in 2017. Their goal is to develop new approaches to spectroscopy that allow them to explore larger and more complex biological structures at the molecular level of the cell.
The next step in this line of research will be to identify other suitable spin labels and to develop RIDME for application in molecules where the distance between the spin labels is unknown. A particular focus of attention will be on molecules associated with neuro-degenerative diseases such as Alzheimer’s and Parkinson’s.
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic World Spectroscopy
Investigation with spectroscopy gives us unique insights into the composition and structure of materials. From UV-Vis spectroscopy to infrared and Raman spectroscopy to fluorescence and atomic absorption spectroscopy, spectroscopy offers us a wide range of analytical techniques to precisely characterize substances. Immerse yourself in the fascinating world of spectroscopy!

Topic World Spectroscopy
Investigation with spectroscopy gives us unique insights into the composition and structure of materials. From UV-Vis spectroscopy to infrared and Raman spectroscopy to fluorescence and atomic absorption spectroscopy, spectroscopy offers us a wide range of analytical techniques to precisely characterize substances. Immerse yourself in the fascinating world of spectroscopy!




















































