Diamond dust shines bright in Magnetic Resonance Imaging
Potential alternative to widely used contrast agent gadolinium
Advertisement
An unexpected discovery surprised a scientist at the Max Planck Institute for Intelligent Systems in Stuttgart: nanometer-sized diamond particles, which were intended for a completely different purpose, shone brightly in a magnetic resonance imaging experiment – much brighter than the actual contrast agent, the heavy metal gadolinium. Could diamond dust – in addition to its use in drug delivery to treat tumor cells – one day become a novel contrast agent used for MRI? The research team now published their discovery in Advanced Materials.
Some of the world’s greatest discoveries happened by accident. While the discovery of diamond dust’s potential as a future MRI contrast agent may never be considered a turning point in science history, its signal-enhancing properties are nevertheless an unexpected finding which may open-up new possibilities: Diamond dust glows brightly even after days of being injected. Does that mean it could perhaps one day become an alternative to the widely used contrast agent gadolinium?
This heavy metal is used in clinics to detect tumors, inflammation, or vascular abnormalities for more than 30 years. It enhances the brightness of the image of affected areas. However, when injected into a patient's bloodstream, gadolinium travels not only to tumor tissue but also to surrounding healthy tissue. It is retained in the brain and kidneys, persisting months to years after the last administration. The long-term effects on the patient are not yet known. Gadolinium also causes a number of other side effects. The search for an alternative has been underway for years.
Could diamond dust, a carbon-based material, become a well-tolerable alternative because of an unexpected discovery made in a laboratory at the Max Planck Institute for Intelligent Systems in Stuttgart?
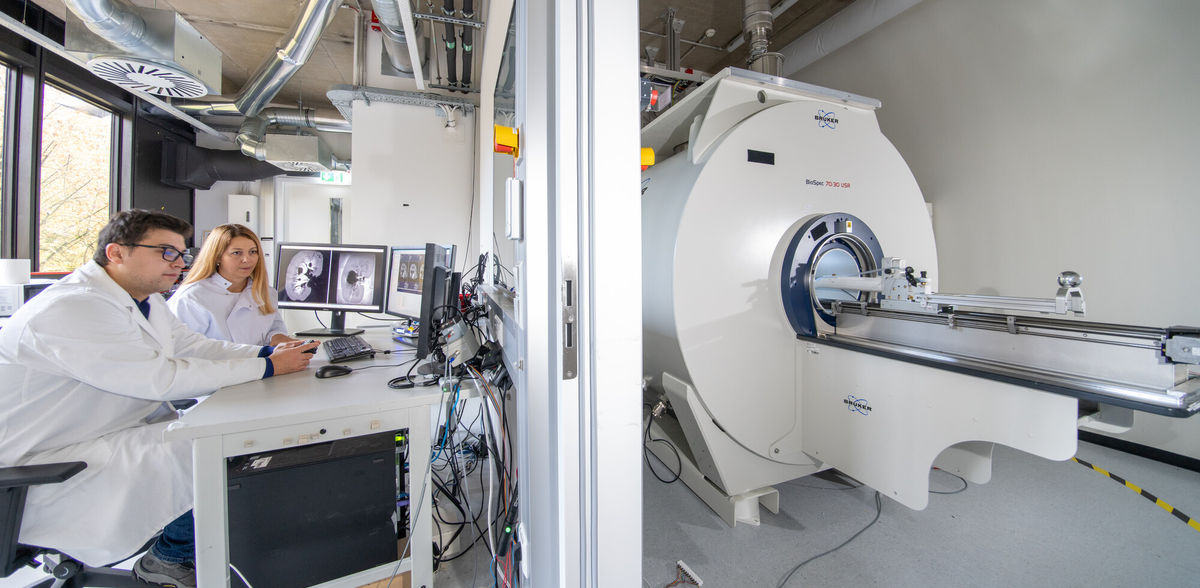
Dr. Jelena Lazovic Zinnanti was working on an experiment using nanometer-sized diamond particles for an entirely different purpose. The research scientist, who heads the Central Scientific Facility Medical Systems at MPI-IS, was surprised when she put the 3 to 5 nanometer particles into tiny drug-delivery capsules made of gelatin. She wanted these capsules to rupture when exposed to heat. She assumed that diamond dust, with its high heat capacity, could help.
“I had intended to use the dust only to heat up the drug carrying capsules,” Jelena recollects. “I used gadolinium to track the dust particles’ position. I intended to learn if the capsules with diamonds inside would heat up better. While performing preliminary tests, I got frustrated, because gadolinium would leak out of the gelatin – just as it leaks out of the bloodstream into the tissue of a patient. I decided to leave gadolinium out. When I took MRI images a few days later, to my surprise, the capsules were still bright. Wow, this is interesting, I thought! The diamond dust seemed to have better signal enhancing properties than gadolinium. I hadn’t expected that.”
Jelena took these findings further by injecting the diamond dust into live chicken embryos. She discovered that while gadolinium diffuses everywhere, the diamond nanoparticles stayed in the blood vessels, didn't leak out and later shone brightly in the MRI, just as they had done in the gelatin capsules. While other scientists had published papers showing how they used diamond particles attached to gadolinium for magnetic resonance imaging, no one had ever shown that diamond dust itself could be a contrast agent.
Two years later, Jelena became the lead author of a paper now published in Advanced Materials.
“Why the diamond dust shines bright in our MRI still remains a mystery to us,” says Jelena, who worked with Prof. Metin Sitti and researchers from the Physical Intelligence Department at MPI-IS and with Dr. Eberhard Goering from the MPI-IS’ neighboring institute, the MPI for Solid State Research. She can only assume the reason for the dust’s magnetic properties: “I think the tiny particles have carbons that are slightly paramagnetic. The particles may have a defect in their crystal lattice, making them slightly magnetic. That’s why they behave like a T1 contrast agent such as gadolinium. Additionally, we don’t know whether diamond dust could potentially be toxic, something that needs to be carefully examined in the future.”
If diamond dust is found to be safe and well tolerated by patients, Jelena believes it has the potential to become a new contrast agent option for future MRI scans, where it would be deposited in tissue with abnormal vasculature, such as tumors, but not in healthy tissue.