The pyrenoid is a carbon-fixing liquid droplet
Advertisement
Plants and algae use the enzyme Rubisco to fix carbon dioxide, removing it from the atmosphere and converting it into biomass. However, Rubisco performs this reaction slowly and can also have unwanted reactions with oxygen. Algae have figured out a clever way to increase the efficiency of carbon fixation. They gather most of their Rubisco into a ball-shaped microcompartment called the pyrenoid, which they flood with a high local concentration of carbon dioxide. If we learn how algae build the pyrenoid, we may be able to engineer it into plants, creating crops that remove more carbon dioxide from the atmosphere while producing more food. Combining genetics, cell biology, computer modeling and cryo-electron tomography, an international team of scientists at Princeton University, the Carnegie Institution for Science, Stanford University and the Max Plank Institute of biochemistry have unravelled the mysteries of how the pyrenoid is assembled. They found that the pyrenoid behaves like a droplet of liquid, which dissolves during cell division to ensure that it is inherited by both daughter cells.
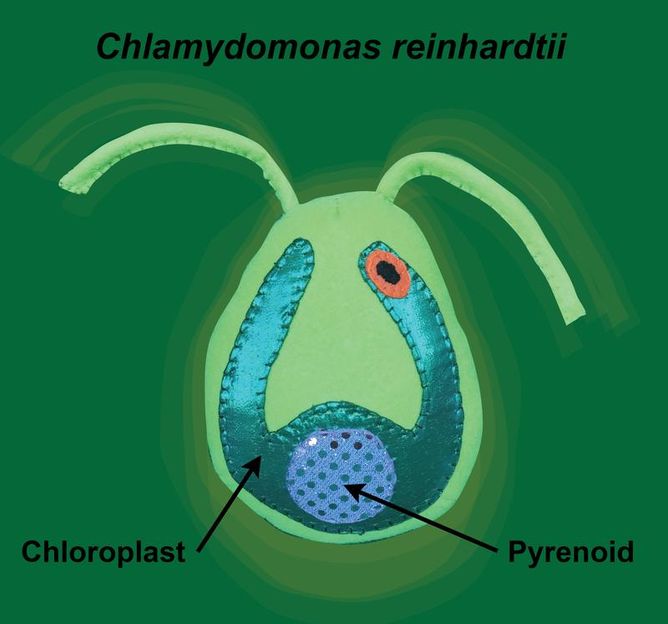
In this image, "Sammy the Chlamy" illustrates how the pyrenoid is a phase-separated microcompartment within the chloroplast of the Chlamydomonas cell.
© Krystal Klaus
A warming planet
Our planet’s climate is changing. Each year brings record high temperatures that cause extreme weather, melting polar ice and rising ocean levels. Global warming is intensified by greenhouse gasses such as carbon dioxide, which prevent the escape of heat from our atmosphere. Using energy from the sun in a process called photosynthesis, plants and algae act as natural air filters, removing carbon dioxide from the atmosphere while replacing it with the oxygen that we breathe. About half of the photosynthesis on Earth is performed in the ocean by single-celled algae. Many of these algae fix carbon dioxide more efficiently than land plants by concentrating most of their Rubisco into a microcompartment called a pyrenoid. Despite the importance of the pyrenoid to the global environment, until recently, it was unknown how this microcompartment assembles.
Visualizing every Rubisco within the pyrenoid
The first breakthrough in understanding pyrenoid assembly came when the team of Martin Jonikas, leader of the Carnegie/Stanford and Princeton groups, identified a linker protein in the green alga Chlamydomonas that binds Rubisco enzymes together within the pyrenoid. Without this “molecular glue,” the pyrenoid does not form. However, it was not known how the Rubisco proteins are organized within the pyrenoid, with classical electron microscopy studies suggesting that the pyrenoid is a highly ordered solid crystal.
To answer this question, the team lead by Benjamin Engel at the Max Planck Institute of Biochemistry used cryo-electron tomography to examine the molecular organization of the pyrenoid within Chlamydomonas cells that were frozen in their native state, avoiding the artefacts caused by sample preparation for classical electron microscopy. This high-resolution imaging technique enabled Engel and colleagues to precisely measure the positions of the thousands of Rubisco enzymes within the pyrenoid. Instead of crystalline organization, they found that the pyrenoid only has short-range order. Engel explains this result: "If you compare our measurements to the organization of molecules inside liquids, there are very clear similarities. This suggests that pyrenoids are actually liquid-like structures."
Like oil and water
In order to prove that the pyrenoid behaves like a liquid, Elizabeth Freeman Rosenzweig, first author of the study, used fluorescence microscopy to measure Rubisco movement within living cells. She used a high-powered laser to destroy the signal from fluorescent labels attached to Rubisco in half of the pyrenoid, while leaving the signal in the other half of the pyrenoid intact. Within minutes, the fluorescence spread throughout the pyrenoid, showing that the enzymes move around as they would in a liquid. Thus, the pyrenoid is a liquid microcompartment floating within a second larger liquid compartment, the chloroplast. This is an example of “phase separation,” a physical phenomenon that has recently been shown to play a role in compartmentalizing many of the cell’s proteins. Freeman Rosenzweig uses an analogy to explain how it works: “Although the forces that cause the pyrenoid’s phase separation are different, it is easy to think about it terms of a familiar image: a dish of oil and vinegar that you might get at an Italian restaurant. The oil and vinegar are both liquids, but they don’t mix. The vinegar instead forms droplets that float in the pool of oil. Similarly, we think the pyrenoid forms a droplet within the liquid environment of the chloroplast.”
Original publication
E.S. Freeman Rosenzweig, B. Xu, L. Kuhn Cuellar, A. Martinez-Sanchez, M. Schaffer, M. Strauss, H.N. Cartwright, P. Ronceray, J.M. Plitzko, F. Förster, N.S. Wingreen, B.D. Engel, L.C.M. Mackinder & M.C. Jonikas; “The Eukaryotic CO2-Concentrating Organelle is Liquid-Like and Exhibits Dynamic Reorganization”; Cell; 2017
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Fluorescence microscopy
Fluorescence microscopy has revolutionized life sciences, biotechnology and pharmaceuticals. With its ability to visualize specific molecules and structures in cells and tissues through fluorescent markers, it offers unique insights at the molecular and cellular level. With its high sensitivity and resolution, fluorescence microscopy facilitates the understanding of complex biological processes and drives innovation in therapy and diagnostics.
Topic world Fluorescence microscopy
Fluorescence microscopy has revolutionized life sciences, biotechnology and pharmaceuticals. With its ability to visualize specific molecules and structures in cells and tissues through fluorescent markers, it offers unique insights at the molecular and cellular level. With its high sensitivity and resolution, fluorescence microscopy facilitates the understanding of complex biological processes and drives innovation in therapy and diagnostics.




















































