Redipor - Ready to use media
Ready prepared culture media for monitoring, testing and validating environments and products
Performance you can trust. Produced under ISO 9001 and ISO/IEC 17025 accreditations
Tailored and fit for purpose. Flexible in formulations, fill volumes, packaging and sterile formats
Reliable supply & support with multi-site production for fast delivery and business continuity
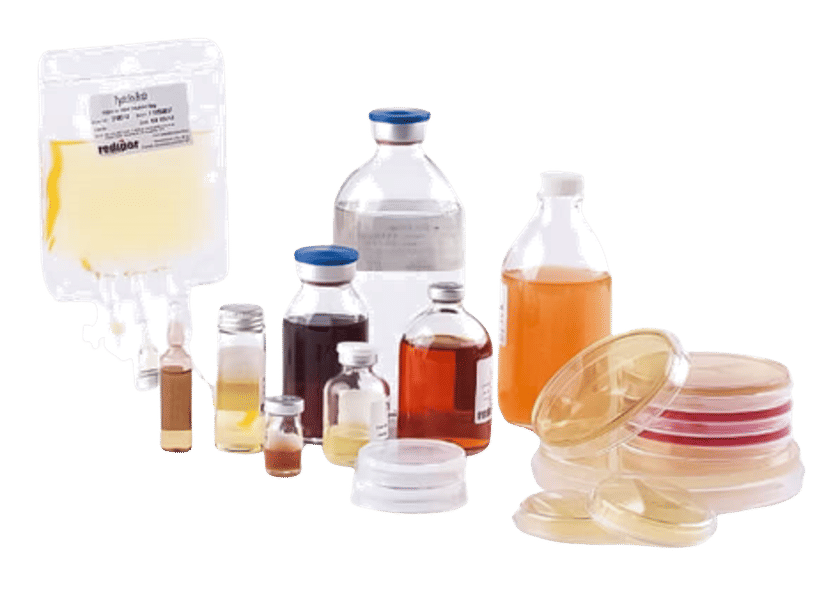






High-quality tailored media solutions for pharma QC, food, beverage, water and clinical diagnostics
Our ready-to-use culture media provide laboratories in pharmaceutical Quality Control, Food and Beverage testing, water testing and clinical diagnostics industries with reliable, efficient, and practical solutions for microbiological analyses. They save valuable time as no preparation is needed and reduce contamination risk with pre-tested, ready-prepared media. This ensures consistent, accurate results that meet the highest quality standards and regulatory requirements.
The flexibility of the media allows them to be seamlessly integrated into diverse laboratory workflows, from routine testing to specialized applications. Whether small batches are needed for specific tests or larger volumes for daily operations, the media support efficient planning and execution of analyses.
Traceability and standardized quality ensure reproducible results and simplify regulatory compliance. In pharmaceuticals, they help detect contamination early and secure the production of safe, compliant products. In food and beverage testing, they enable rapid pathogen detection, hygiene monitoring, and process validation to protect consumers and maintain product quality.
For water testing, the media provide a reliable basis for detecting harmful microorganisms and ensure compliance with regulatory standards, supporting monitoring of industrial and environmental water systems. Laboratories can perform even demanding tests quickly and accurately.
Additionally, ready-to-use media simplify laboratory operations by reducing preparation time, equipment needs, and staff training requirements. This minimizes errors and contamination risk while improving productivity and workflow efficiency.
In summary, these culture media deliver safe, reproducible results, boost efficiency, reduce risks, and help laboratories reliably meet stringent regulatory and quality standards. They are the ideal solution for labs that demand precise, safe, and efficient microbiological testing.

1
Redipor by AnalytiChem - Product range

2
Ensuring Pharmaceutical Safety with Redipor sterility testing

3
Microbial Testing is Critical for Food Safety

4
Water testing

5
Isolating and Identifying Clinical Pathogens
Request information about Redipor - Ready to use media now

Culture media: Redipor - Ready to use media
Ready prepared culture media for monitoring, testing and validating environments and products


