FDA Approves Avastin in Combination with Chemotherapy for Second-Line Metastatic Colorectal Cancer Patients
Advertisement
Genentech, Inc. announced that the U.S. food and Drug Administration (FDA) approved Avastin® (bevacizumab) in combination with intravenous 5-fluorouracil (5-FU)-based chemotherapy for second-line metastatic colorectal cancer. Avastin is also approved as a first-line treatment of metastatic colorectal cancer (CRC) in combination with intravenous 5-FU-based chemotherapy.
"Avastin is the only biologic therapy with a demonstrated survival benefit in colorectal cancer, and this new indication offers CRC patients who have received a previous treatment regimen a new option to help fight their disease," said Hal Barron, M.D., senior vice president, Development and chief medical officer at Genentech. "Avastin used in combination with chemotherapy has become an important component of care for patients with metastatic colorectal cancer. We continue to study Avastin in both the adjuvant and metastatic settings, and Phase III trials in kidney, breast, pancreatic, non-small cell lung, prostate and ovarian cancers are ongoing in the hope that we may be able to help patients with other cancers as well."
The approval is based on results of a randomized, controlled, multicenter Phase III trial (E3200) of 829 patients with advanced or metastatic CRC who had received previous treatment with irinotecan and 5-FU as initial therapy for metastatic disease or as adjuvant therapy. The study showed that patients who received Avastin plus the 5-FU-based chemotherapy regimen known as FOLFOX4 (oxaliplatin/5-FU/leucovorin) had a 25 percent reduction in the risk of death (based on a hazard ratio of 0.75), the primary endpoint, which is equivalent to a 33 percent improvement in overall survival, compared to patients who received FOLFOX4 alone. Median survival for patients receiving Avastin plus FOLFOX4 was 13.0 months, compared to 10.8 months for those receiving FOLFOX4 alone.
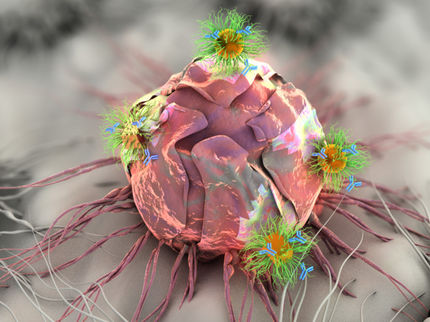
Avastin is a therapeutic antibody designed to inhibit Vascular Endothelial Growth Factor (VEGF), a protein that plays an important role in tumor angiogenesis and maintenance of existing tumor vessels. By inhibiting VEGF, Avastin is designed to interfere with the blood supply to a tumor, a process that is thought to be critical to a tumor's growth and metastasis.