OctoPlus announces positive Phase IIa efficacy and tolerability results for Locteron in hepatitis C
Advertisement
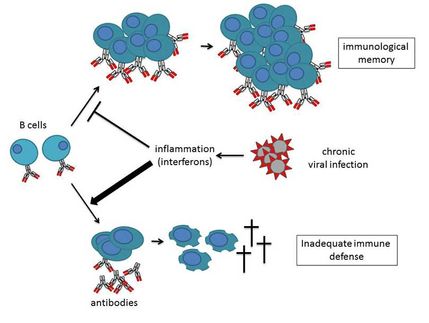
OctoPlus N.V. announced the successful completion of the Phase IIa clinical study with its lead product Locteron(TM), a controlled release interferon alfa product for the treatment of chronic hepatitis C (HCV).
According to the company, results from this study confirm Locteron's potential to substantially improve patient care in HCV. Safety data from the study show a substantially improved tolerability profile for Locteron compared to other interferon products on the market or in development. Efficacy data from the study indicate Locteron's strong antiviral effect, with 100% of the patients achieving early virologic response in the two highest dose groups. In addition, Locteron is a more convenient therapy than existing treatments due to its controlled-release profile, allowing for once every two weeks drug administration versus the current once a week regimen. OctoPlus is co-developing Locteron with its partner Biolex Therapeutics.
Locteron is designed to be a best-in-class therapeutic for patients with chronic hepatitis C, with the potential to induce less side effects, improve patient compliance and provide a more convenient once every two week dosing schedule compared with current therapies. Locteron combines OctoPlus' proprietary PolyActive(TM) drug delivery technology with BLX-883, a recombinant interferon alfa produced by OctoPlus' co-development partner Biolex Therapeutics in its patented LEX System(SM). Locteron is produced in OctoPlus' cGMP manufacturing facilities in Leiden, the Netherlands.