Wyss Institute at Harvard University launches SPEAR Bio
Spear Bio is working towards commercializing its first assay, which accurately assesses levels of SARS-CoV-2-neutralizing antibodies in dried-blood spot samples
Advertisement
The Wyss Institute for Biologically Inspired Engineering at Harvard University and Spear Bio, Inc. announced that the Institute’s DNA nanotechnology-driven ultrasensitive SPEAR protein-detection technology has been licensed to the newly formed Boston-based startup. Spear Bio has signed a worldwide, exclusive licensing agreement with Harvard’s Office of Technnology Development (OTD), granting SPEAR Bio the rights to commercialize the SPEAR protein detection technology. Spear Bio will develop a reagent-based platform for ultrasensitive protein detection in small-volume samples with an initial focus on research-use-only applications.

Symbolic image
pixabay.com
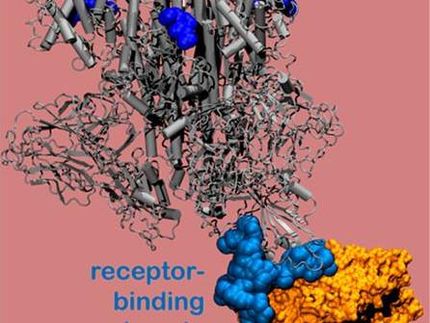
Pioneered in the group of Wyss Core Faculty member Peng Yin, Ph.D., the SPEAR technology enables ultrasensitive protein detection in small patient samples, such as a drop of blood from a finger-stick, dried blood spot samples, and other biofluids obtained with the help of micro-sampling techniques, while leveraging existing laboratory equipment, including the now ubiquitous qPCR machines. Spear Bio plans to use the SPEAR technology to build a more broadly applicable protein detection platform, and will first focus on commercializing an ultra-sensitive assay that can accurately measure the levels of neutralizing antibodies (NAbs) against SARS-CoV-2.
The generation and levels of NAbs are a key metric for understanding protective immunity and vaccine efficacy, and SARS-CoV-2 NAb-detecting assays are tools used by the Centers for Disease Control, as well as vaccine and drug developers, to determine the susceptibility of individuals to infectious pathogens such as COVID-19. The ability to sensitively and accurately quantify them in small, easy-to-obtain patient samples could significantly increase the depth and throughput of such studies, and enable various types of future research and diagnostic assays.
“The invention of SPEAR was enabled by key advances in DNA nanotechnology that we made at the Wyss over the years, including the prescribed and signal-dependent synthesis of readable DNA sequences,” said Peng Yin, Ph.D., who is a lead of the Wyss Institute’s Molecular Robotics Initiative and a Professor of Systems Biology at Harvard Medical School (HMS). “The detection platform that Feng Xuan built and then substantially de-risked with other members in the lab, now has significant potential for developing immunoassay products for clinical research and in vitro diagnostics in the near-term.” Yin, who co-founded Spear Bio, previously co-founded other startups, including Ultivue Inc., NuProbe Global, Torus Biosystems Inc., 3EO Health, and Digital Biology, Inc., which leverage technologies developed in his Wyss Institute lab.
Feng Xuan, Ph.D., was a Postdoctoral Fellow on Yin’s team and also became a co-founder of SPEAR Bio and is now the company’s CTO. During the technology development, he worked with co-inventors Cherry (Tsz Wing) Fan, Ph.D., and Yu Wang, Ph.D., and other members of the group. Wang now is SPEAR Bio’s Acting Head of Application Development.
In SPEAR, which stands for “Successive Proximity Extension Amplification Reaction,” miniscule amounts of proteins, including NAbs, can be detected via target-binding probes that bind to different but proximal sites in a protein’s structure. This proximal double-tagging event allows the two probes to “shake hands,” with their interaction triggering a specifically engineered successive extension reaction, and synthesis of a unique DNA sequence which then can be amplified and quantified using standard qPCR instruments. Importantly, in the absence of detection targets, the interaction between free-floating probes does not allow the synthesis of the complete DNA sequence, significantly reducing the background compared to conventional proximity-based assays. SPEAR is superior to other protein detection assays in its combination of extreme sensitivity, wash-free workflow, and functionality over a large range of target protein levels (dynamic range) with the ability to be fully effective in sample volumes as small as 1 uL. The technology was de-risked with the help of the Wyss’ translation engine, in which it received the status first of a Validation Project, and then of an Institute Project, designated to support the development of high-value technologies with high potential for market success.
“The extreme sensitivity in very small sample volumes provided by SPEAR, and the fact that it can be read out using common quantitative PCR equipment, offer unique potential for creating microsampling-based in vitro diagnostics that can transform academic and clinical research in multiple disease areas,” said Xuan.
Spear Bio is presently applying the assay to the quantification of SARS-CoV-2 NAb in dried blood samples, and thereby aims to facilitate COVID-19 disease and vaccine research. Beyond this first application, the company plans to use the assay to develop other research and diagnostics applications that require the ultrasensitive and quantitative detection of protein biomarkers in small samples. “SPEAR’s unique capabilities give us a clear value proposition for market entry.” said Oliver Tassinari, Sr. Director of Business Development at SPEAR Bio. “We are now laser-focused on translating the exceptional technical performance of the assay technology to a satisfying customer experience to solve research and diagnostics related problems.”
Harvard initially granted SPEAR Bio access to this technology on a non-exclusive basis, for a limited term, in accordance with the University’s commitment to the COVID-19 Technology Access Framework. The Framework enables broad access to emerging technologies to incentivize rapid innovation in pursuit of solutions to combat the pandemic.
“Our ability to detect ever smaller amounts of biomolecules across all molecular species with increasing speed and specificity, and in various settings including dried blood samples, opens entirely new approaches to medical diagnostics that could be used at home as well as in hospital settings. The Wyss’ SPEAR protein detection technology, developed by Peng Yin’s group, is at the forefront of this field, and should help move the needle in terms of rapid clinical assessment of patient protection after vaccination or infection during COVID-19 in the near term,” said Wyss Founding Director Donald Ingber, M.D., Ph.D., who is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children’s Hospital, and the Hansjörg Wyss Professor of Bioinspired Engineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.
Other news from the department business & finance
Most read news
More news from our other portals
Something is happening in the life science industry ...
This is what true pioneering spirit looks like: Plenty of innovative start-ups are bringing fresh ideas, lifeblood and entrepreneurial spirit to change tomorrow's world for the better. Immerse yourself in the world of these young companies and take the opportunity to get in touch with the founders.
See the theme worlds for related content
Topic world Protein analytics
Protein analytics provides a deep insight into these complex macromolecules, their structure, function and interactions. It is essential for discovering and developing biopharmaceuticals, understanding disease mechanisms, and identifying therapeutic targets. Techniques such as mass spectrometry, Western blot and immunoassays allow researchers to characterize proteins at the molecular level, determine their concentration and identify possible modifications.

Topic world Protein analytics
Protein analytics provides a deep insight into these complex macromolecules, their structure, function and interactions. It is essential for discovering and developing biopharmaceuticals, understanding disease mechanisms, and identifying therapeutic targets. Techniques such as mass spectrometry, Western blot and immunoassays allow researchers to characterize proteins at the molecular level, determine their concentration and identify possible modifications.