Wyatt Technology
The Diffusion Interaction Parameter (kD) as an Indicator of Colloidal and Thermal Stability

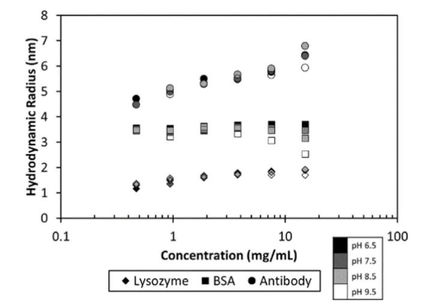
Stability is a key quality attribute in formulation studies of po-tential therapeutic biomolecules. In order to minimize time, effort and funds spent on stability studies, researchers rely on high-throughput screening methods that can reliably test hundreds of combinations of candidates, excipients and buffer conditions. Techniques utilized in these screens must determine a variety of stability-indicating parameters (SIPs). Some of the most useful SIPs to date are short term aggre-gation, thermal stability; and colloidal stability.
The ability to screen protein formulations at the early stages of development enables scientists to concentrate on the most suitable candidates, therefore saving substantial amounts of time, sample and testing equipment. This experiment demonstrates that thermal and colloidal stability of proteins, two indicators of propensity to aggregate, as well as actual aggregation states, are all determined simultaneously during the screening process with dynamic light scattering (DLS) tools in order to rank the effectiveness of candidates and for-mulation conditions. DLS can also indicate chemical stability and the average molar mass and specific volume of mole-cules in solution. For these reasons, HTS-DLS provides sub-stantial quantities of information for the rapid screening of candidate molecules, buffer conditions and excipients, allow-ing the DynaPro® Plate Reader III to maximize productivity in formulation studies.
Advertisement
White Paper classification
White papers on related topics
Products on related topics
See the theme worlds for related content
Topic world Protein analytics
Protein analytics provides a deep insight into these complex macromolecules, their structure, function and interactions. It is essential for discovering and developing biopharmaceuticals, understanding disease mechanisms, and identifying therapeutic targets. Techniques such as mass spectrometry, Western blot and immunoassays allow researchers to characterize proteins at the molecular level, determine their concentration and identify possible modifications.

Topic world Protein analytics
Protein analytics provides a deep insight into these complex macromolecules, their structure, function and interactions. It is essential for discovering and developing biopharmaceuticals, understanding disease mechanisms, and identifying therapeutic targets. Techniques such as mass spectrometry, Western blot and immunoassays allow researchers to characterize proteins at the molecular level, determine their concentration and identify possible modifications.