Turning harmful carbon dioxide into useful chemicals
Inspired by nature: Electro-biocatalytic CO₂ fixing replaces fossil resources
Advertisement
Making important raw materials for fine chemicals out of carbon dioxide really works. As part of the Max Planck collaborative project eBioCO2n, a team of researchers from Fraunhofer IGB have successfully performed a first ever fixation of CO2 via a multi-enzyme enzyme reaction driven by electricity yielding a prospective intermediate for the chemical industry. The process for electro-biocatalytic CO2 fixation was recently published and is considered a “hot paper.”

Enzyme reactions are combined for the process: one for provision and regeneration of the cofactor, one for CO2 fixation.
© Fraunhofer/Marc Müller
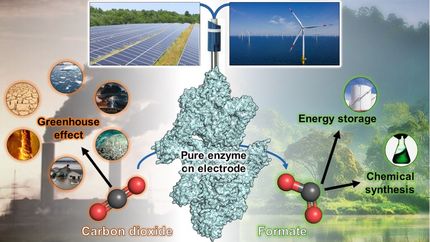
Burning fossil resources produces carbon dioxide, a greenhouse gas that is harmful to the climate and contributes significantly to global warming. Fossil resources, however, remain a key resource – not just as a source of energy, but as a raw material for the chemical industry that is used in many daily essentials such as medicines, packaging, textiles, cleaning products and more. Intensive research is therefore being conducted into a number of alternatives to fossil resources.
Renewable resources are one promising option, but other alternative resource bases are also required if the availability of green synthetics is to keep up with needs in the years to come. The ability to fix CO2 in a targeted way under mild reaction conditions is another sustainable driver of a circular carbon economy.
Carbon capture from the air for lower CO2 emissions
A team of researchers at the Straubing institute branch of the Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB, together with colleagues from the Max Planck Institute for Terrestrial Microbiology in Marburg and the Technical University of Munich, has successfully performed a first ever bioelectrocatalytic conversion of CO2 into valuable substances for the chemical industry. By combining different approaches from bioelectrochemistry, enzyme biology and synthetic biology, special bioelectrodes have been developed to use renewable energy to drive enzymes to produce valuable organic molecules from the greenhouse gas in a sequential reaction similar to photosynthesis.
The aim is to capture CO2 directly from the air: “This would allow the process not only to help industry to move away from fossil resources, but also to actively drive the climate transition by reducing CO2,” explains Dr. Michael Richter, Head of Innovation Field Bioinspired Chemistry at Fraunhofer IGB. “First of all, though, we needed to show that our idea of driving such a complex biocatalytic multi-enzyme reaction with electricity like this would work at all.”
Hydrogel transports electrons for CO2-fixing enzymes
Their efforts were successful: The metabolic processes of microorganisms inspired the researchers to develop an electricity-based process for CO2 fixing. The main elements in this process are CO2-fixing enzymes developed by Dr. David Adam and Prof. Tobias Erb, Director of the MPI in Marburg. The next challenge was to provide the CO2-fixing enzymes with a continuous supply of the electrons needed for the reduction of CO2. This was achieved by embedding the enzymes in a redox-active hydrogel, allowing them to be electrochemically driven and bind carbon dioxide to a substrate, thus converting it into a valuable intermediate. “The process is a highly efficient reaction path, reductive carboxylation, that is highly economical and clean because it doesn’t require the presence of any other substances in the system – just carbon dioxide, substrate and electrons, preferably from renewable sources,” explains Dr. Leonardo Castañeda-Losada, whose doctoral thesis was dedicated to the topic of bioelectrocatalysis and who is now working on the project at Fraunhofer IGB together with Dr. Melanie Iwanow and Dr. Steffen Roth.
The hydrogels in which the enzymes do their work were specially developed at TU Munich under Prof. Nicolas Plumeré. They have been modified to be good conductors of electrons while also offering optimal working conditions for the biomolecules. “This allows us not only to apply monolayers to enzymes, but also to expand this three-dimensionally many times over, because the electrons are conducted everywhere in the gel. Because of this, the process is likely to be readily scalable for the chemical industry in the future,” explains Prof. Volker Sieber, who has been pursuing CO2 storage strategies at the Straubing institute branch of Fraunhofer IGB for a long time now.
Cofactors are permanently regenerated while the process is running
The completely new approach taken by the researchers depends not only on the ability to drive an enzymatic reaction sequence successfully using electricity, but involves a highly innovative module: For the reactions to take place as intended and ultimately lead to the highest possible product yield, a continuous “doping” feed for the enzyme is needed in the form of suitable functional cofactors. These small organic molecules are used up in the course of each reaction and need to be regenerated in order to be used again. Not regenerating them is very costly and therefore uneconomical for the industry. That is why the eBioCO2n experts have found a way to regenerate them, in theory an unlimited number of times, within the same reaction systems in the hydrogels by using electricity. “A cofactor should really only need to be fed into the system once in order to be regenerated automatically again and again. In practice, though, it doesn’t work quite as well as that because the cofactor does not remain stable indefinitely – but it does last a very long time,” says Richter.
The researchers have a whole set of different enzymes available to them for the bioelectrocatalytic cofactor recycling process, discovered through from different organisms. The spectrum of these biomolecules could be extended on a modular basis for additional work, depending on the application, and used as a platform system. “Practically any enzyme could be selected from bioinformatic databases, manufactured by biotechnological means and incorporated into the hydrogels,” says Richter. “This could conceivably allow different biobased fine chemicals to be produced. These fine chemicals could then be expanded through further enzyme cascades so they could practically be diversified as much as necessary.” This is where the expertise of the Marburg MPI in particular comes in. If this can be scaled accordingly, the platform technology could become a promising business model for the chemical industry.
Platform system to be expandable and scalable without limitation
With bioinspired laboratory CO2 fixation, Fraunhofer IGB could carboxylate a coenzyme A derivative, a biomolecule that is important in many metabolic processes. “This has so far been the most challenging molecule to fix CO2 to by biocatalytic means,” says Richter. “Using this technology to modify such a large and structurally sophisticated substance is no small matter.” The latest challenge for the researchers is now to prove that their idea works in a reliable and scalable way and can be expanded on a modular basis. The mood at Fraunhofer IGB is optimistic, however, particularly in light of a well functioning interdisciplinary team, the researcher emphasizes. Industry partners will be brought in for follow-up projects as soon as possible.